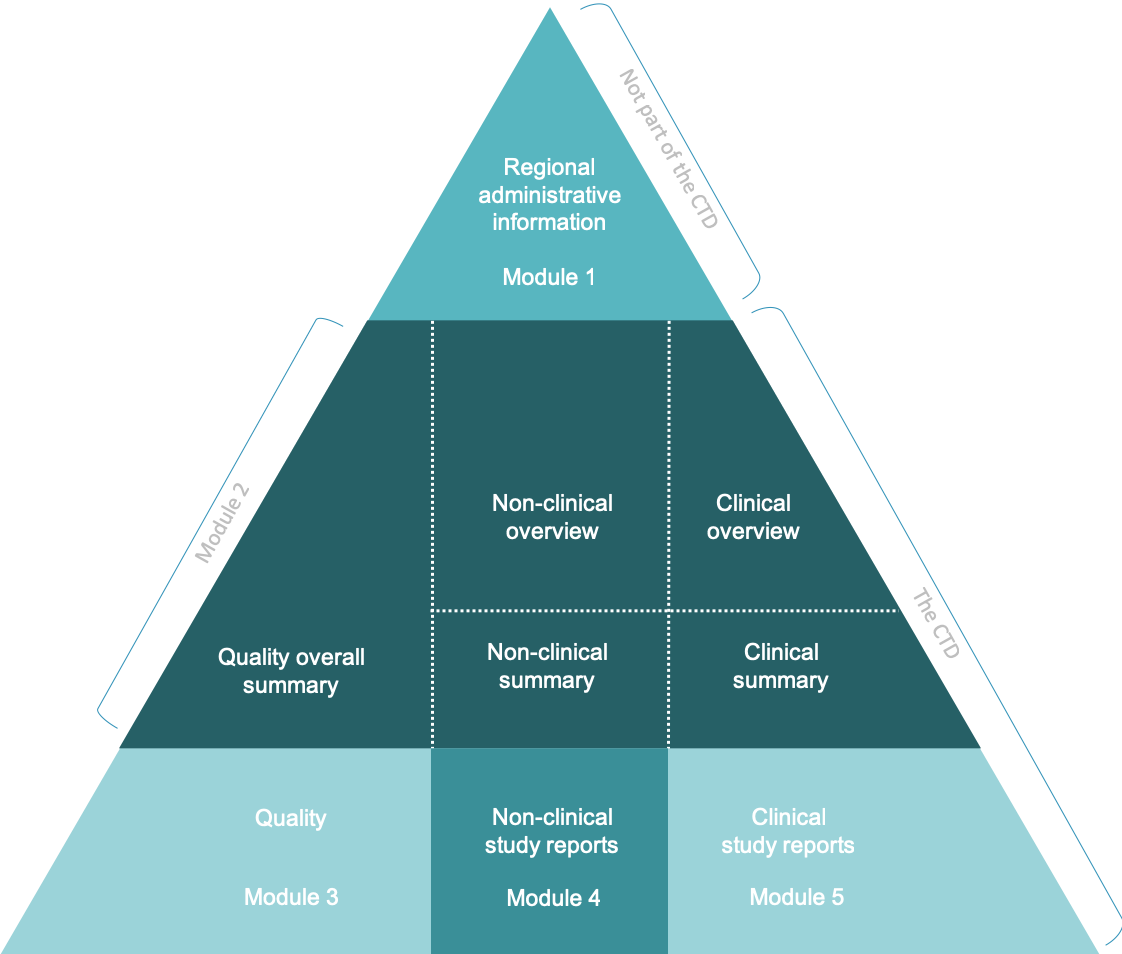
Dossier System as a Practical Tool for Compiling Reimbursement Lists - Value in Health Regional Issues

Proposed clinical trial application dossier. AMPs auxiliary medicinal... | Download Scientific Diagram

What you Need and When – The Key Documents in the Drug Lifecycle - Trilogy Writing & Consulting GmbH

Information on new drugs at market entry: retrospective analysis of health technology assessment reports versus regulatory reports, journal publications, and registry reports | The BMJ
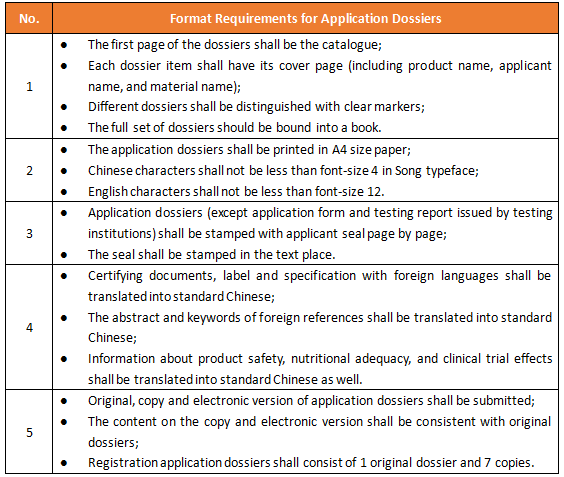
Dossier Requirements of Foods for Special Medical Purpose (FSMP) Registration in China - Regulatory News - Food & Food Contact Materials - CIRS Group
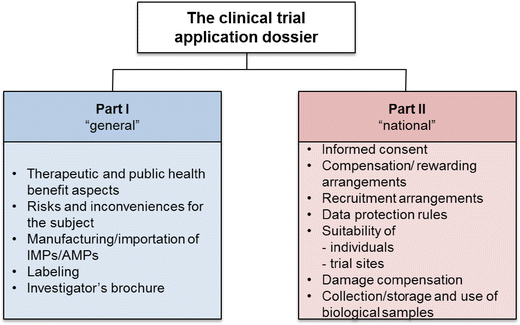
Will the EU Clinical Trials Regulation Support the Innovative Industry in Bringing New Medicines Faster to Patients? | SpringerLink


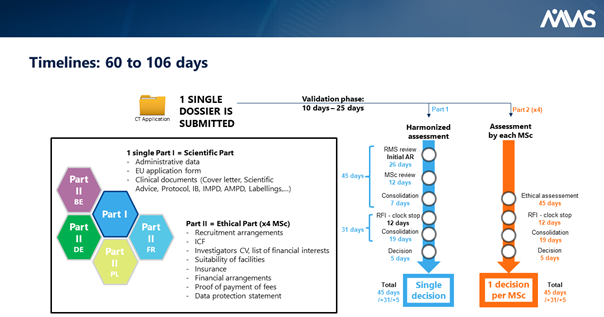






.png)