Whitehall Training - Want a handy guide explaining GCP guidelines and recent changes? Here's a snapshot of the GCP principles including recent amendments! Click the link to buy now: goo.gl/bK42I2 | Facebook

More than a box to check: Research sponsor and clinical investigator perspectives on making GCP training relevant - ScienceDirect
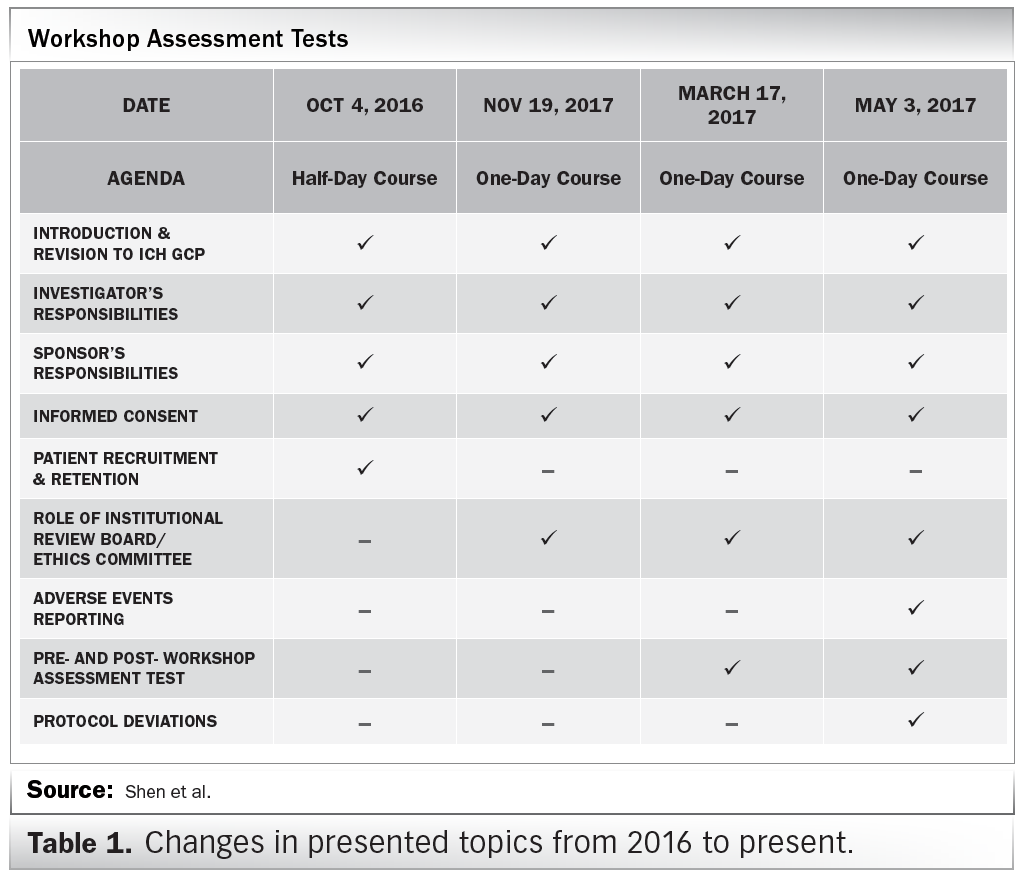
![PDF] The importance of Good Clinical Practice guidelines and its role in clinical trials | Semantic Scholar PDF] The importance of Good Clinical Practice guidelines and its role in clinical trials | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/7a90efbaf9d59ee20353e700230ccbf1660f16b5/3-Table2-1.png)
PDF] The importance of Good Clinical Practice guidelines and its role in clinical trials | Semantic Scholar

EU clinical research framework. ICH GCP = International Conference on... | Download Scientific Diagram

A GCP Primer: Understanding the Basic Application of Good Clinical Practice in the Regulated Environment for the Quality Professional | IVT - GCP
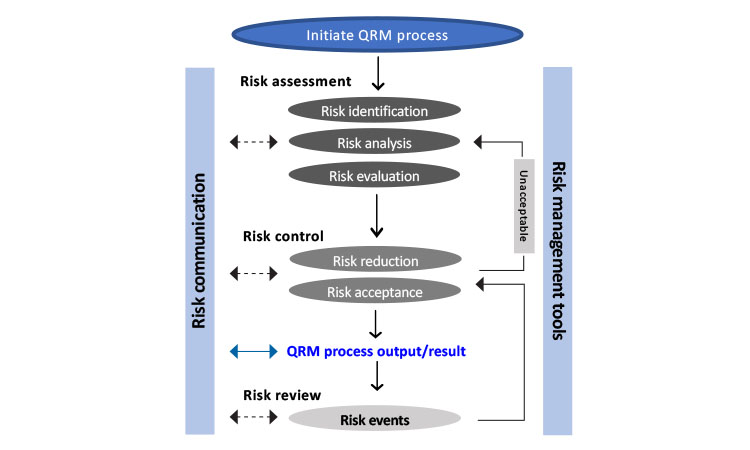
Clinical Trial Management Adaptation to ICH E6 (R2): Good Clinical Practice | Pharmaceutical Engineering
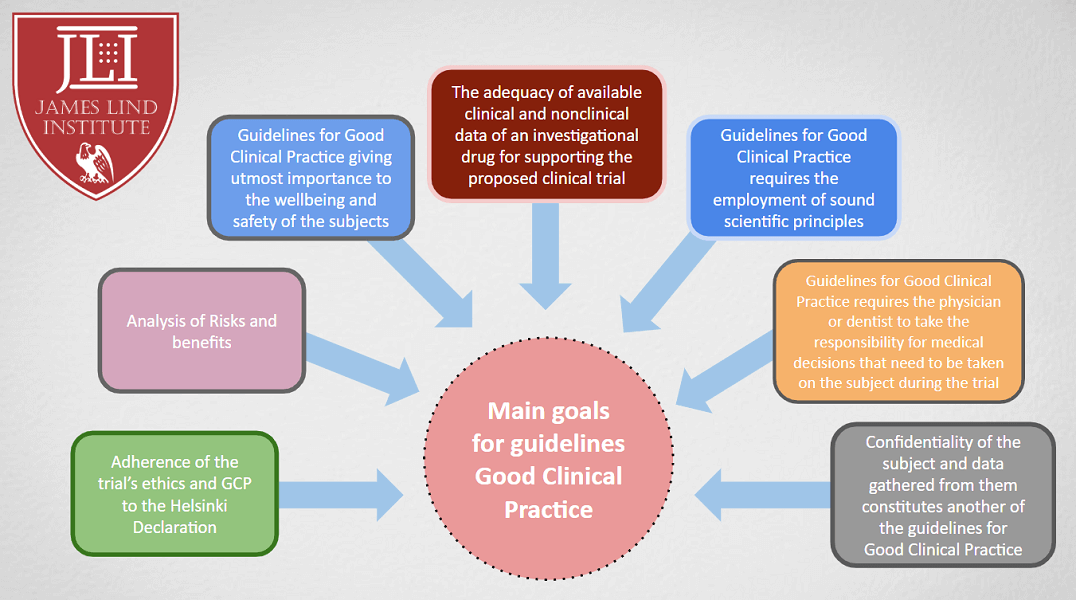
![PDF] Good clinical practice (GCP): A universal call for ethics in biomedical research | Semantic Scholar PDF] Good clinical practice (GCP): A universal call for ethics in biomedical research | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/0fa5ef83b7fba10f4226038b80abe695b3ef999a/4-Figure1-1.png)
PDF] Good clinical practice (GCP): A universal call for ethics in biomedical research | Semantic Scholar