
REFRESHER: ICH Good Clinical Practice (GCP) E6 (R2) and regulatory requirements for Clinical Trials (Fiona Stanley Hospital) - RETProgram

Guideline for good clinical practice E6(R2) / guideline-for-good-clinical- practice-e6-r2.pdf / PDF4PRO

Multi-Regional Clinical Trials Center on Twitter: "The MRCT Center, w/@PartnersCPD, has developed a free, online course reflecting @ICH_news training on ICH E6(R2): Guideline for Good Clinical Practice (GCP).This course is an ICH

ICH GCP Guidelines E6 Revision, R2 Addendum - Changes Impacting Sponsors-CRO-Sites - Webinar Compliance

Introduction To Investigators Responsibilities With Good Clinical Practice | PDF | Institutional Review Board | Clinical Trial

Book M1: 2021 Mini Pocket-Sized (3" x 5") ICH Guidelines for GCP (E6) – Clinical Research Resources, LLC

ICH Public Web Conference on ICH E6 Guideline for Good Clinical Practice - Update on Progress | Pharmaceuticals and Medical Devices Agency

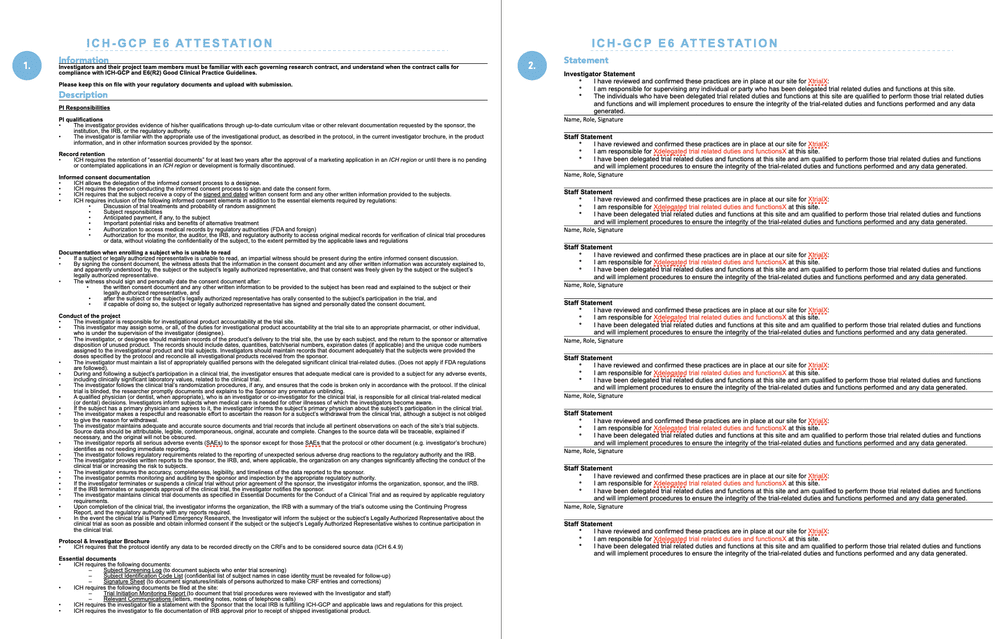














.jpg)

