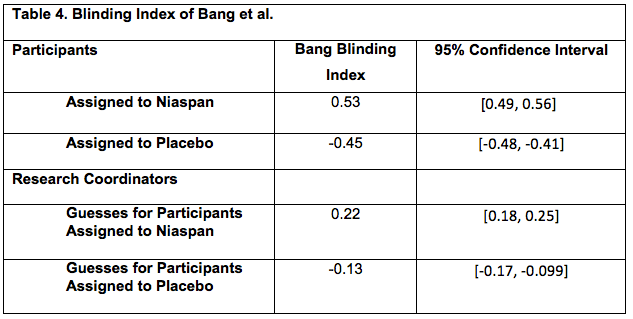
Clinical trial registration was associated with lower risk of bias compared with non-registered trials among trials included in systematic reviews - Journal of Clinical Epidemiology

Masking in Pragmatic Trials: Who, What, and When to Blind - Jennifer B. Christian, Emily S. Brouwer, Cynthia J. Girman, Dimitri Bennett, Kourtney J. Davis, Nancy A. Dreyer, 2019

Descriptors of blinding/masking in clinical trial registries other than... | Download Scientific Diagram
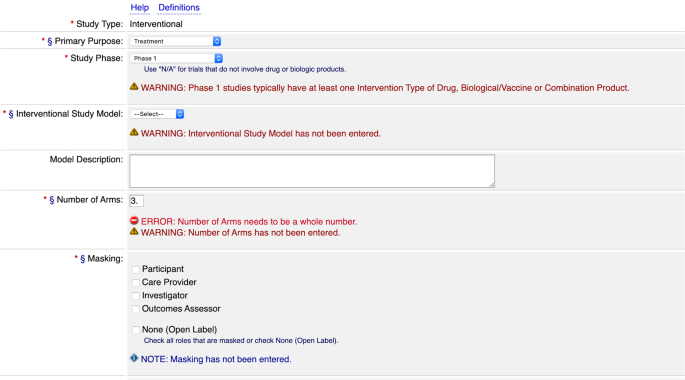
MSM Institutional Review Board MSM IRB Glossary of Clinical Trial Terms http://clinicaltrials.gov/ct2/info/glossary 2/13

Descriptors of blinding/masking in clinical trial registries other than... | Download Scientific Diagram
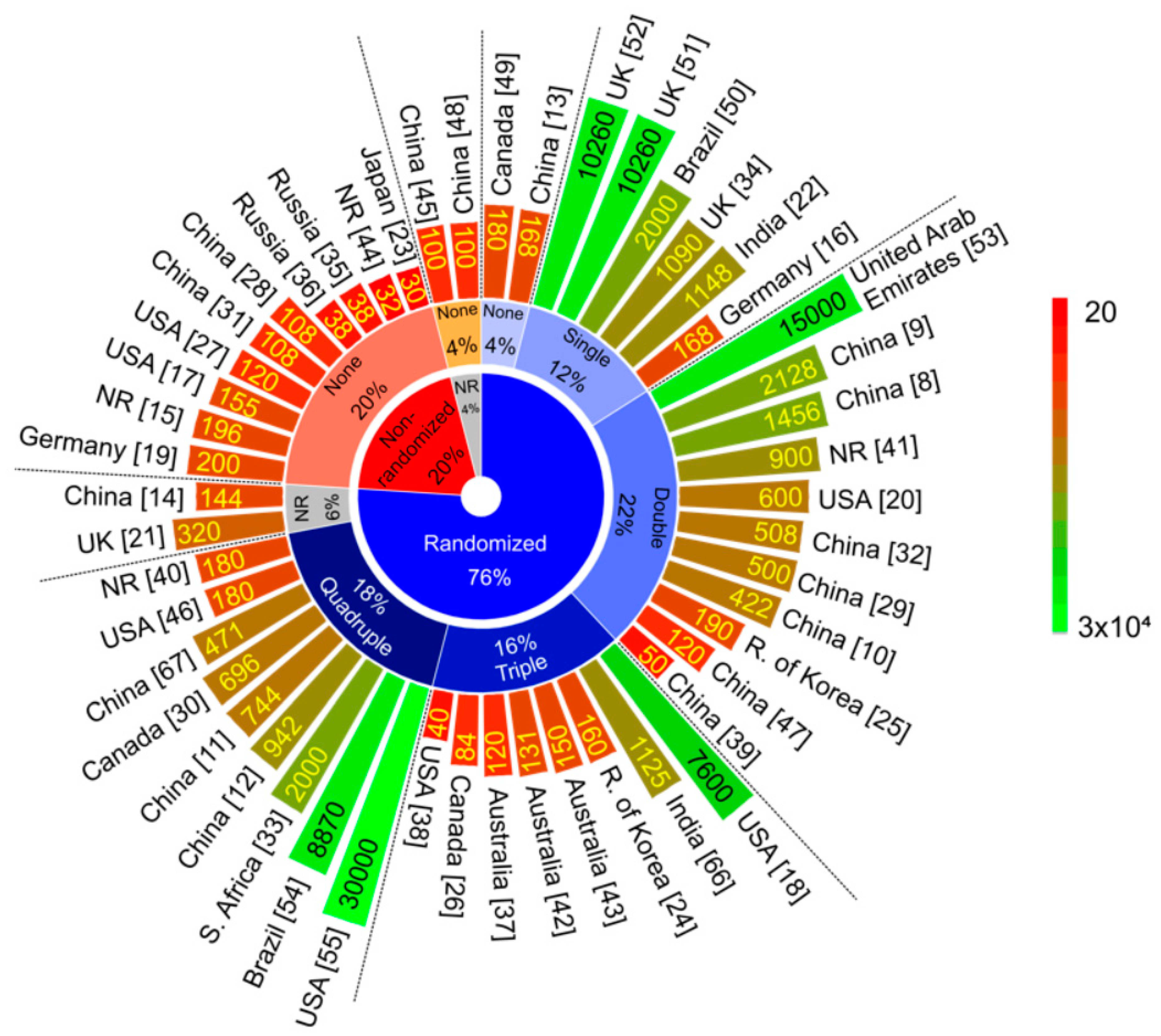
Vaccines | Free Full-Text | Current Clinical Trials Protocols and the Global Effort for Immunization against SARS-CoV-2
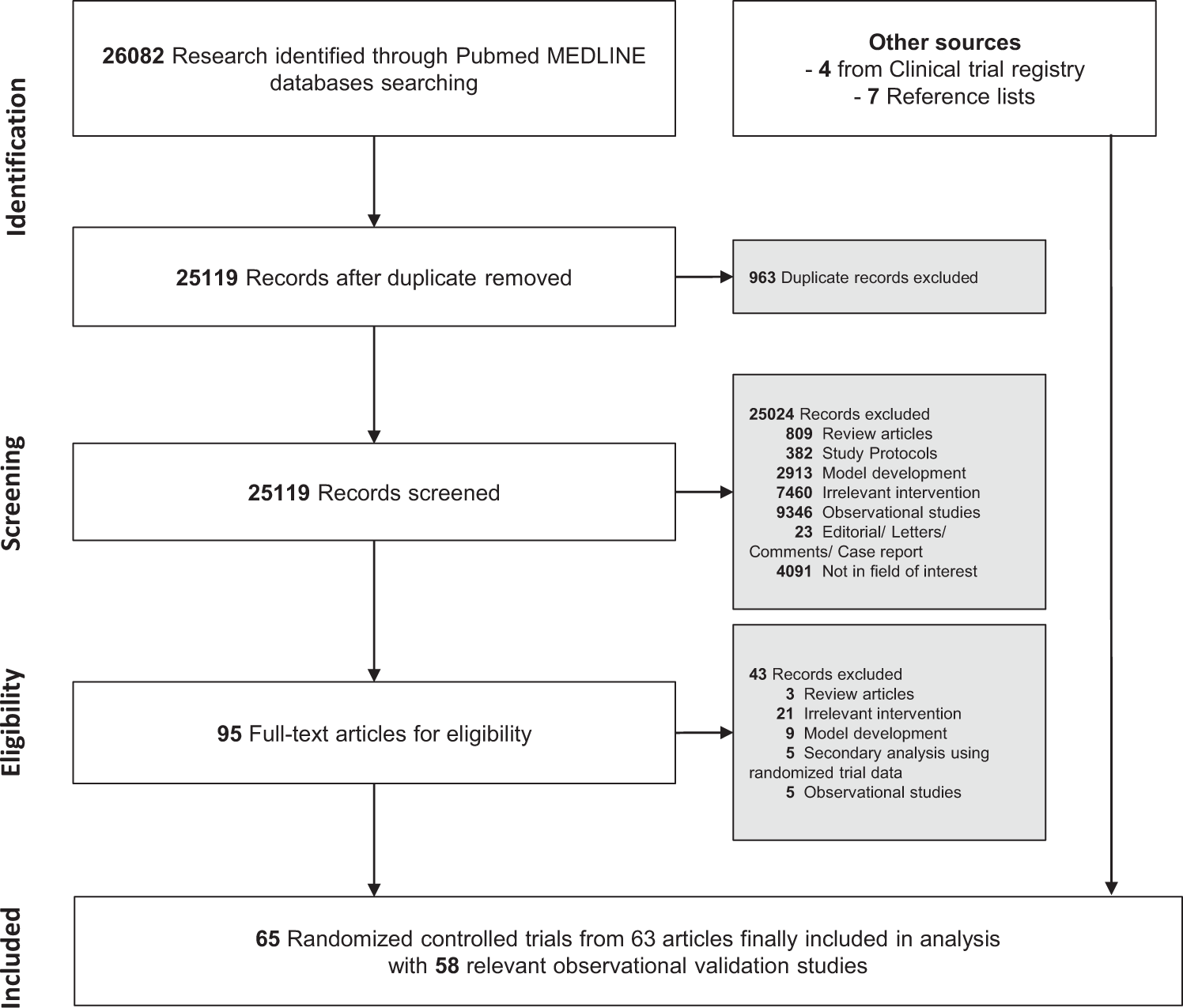
Clinical impact and quality of randomized controlled trials involving interventions evaluating artificial intelligence prediction tools: a systematic review | npj Digital Medicine

Jeremy Howick - 'Blinding' or 'masking' in clinical trials is usually but not always a good thing. See the chapter from my book 'Questioning Double Blinding as a Universal Methodological Virtue of