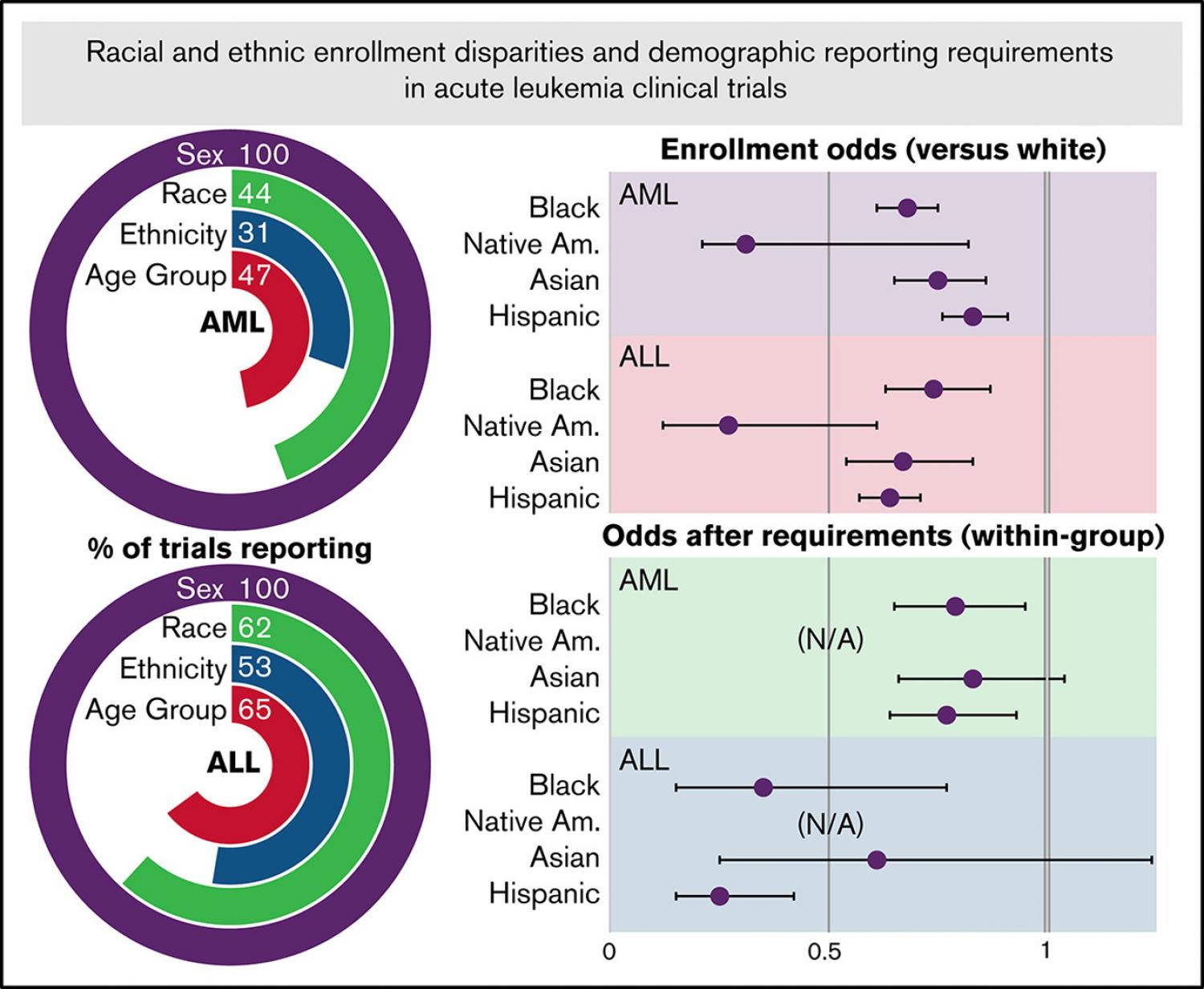
Racial and ethnic enrollment disparities and demographic reporting requirements in acute leukemia clinical trials | Sustainable Development Goals - Resource Centre

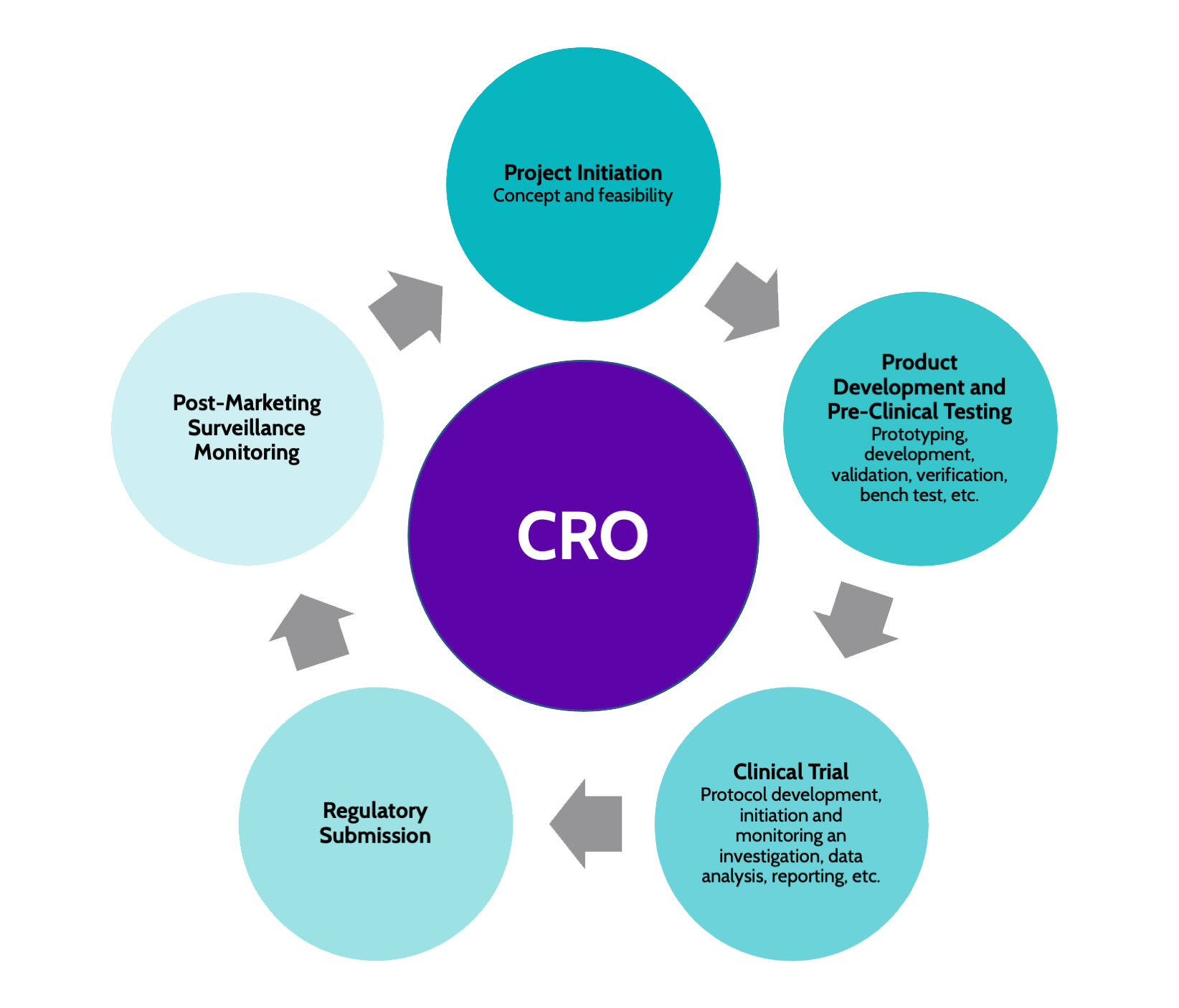
1 Nuts and Bolts of Safety Reporting The Role of the CRO Dr. Noa Lowenton Spier Pharma-Clinical S.A.G. - ppt download
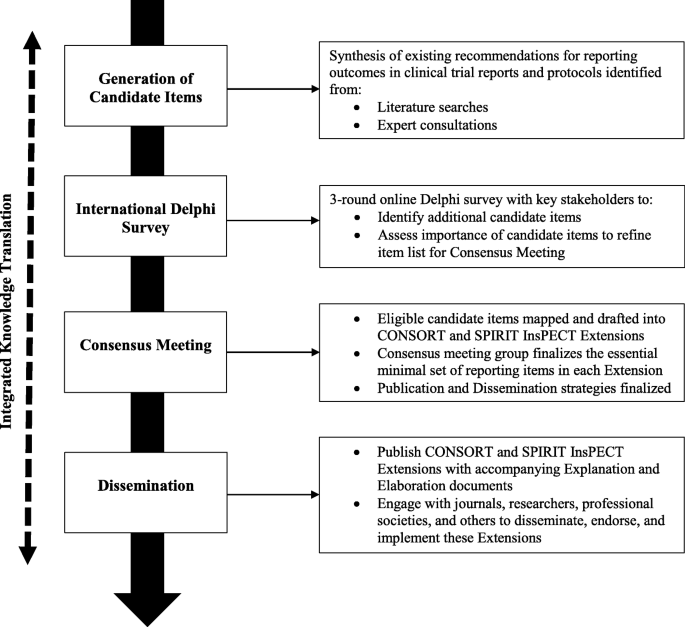
Improving outcome reporting in clinical trial reports and protocols: study protocol for the Instrument for reporting Planned Endpoints in Clinical Trials (InsPECT) | Trials | Full Text

Compliance with legal requirement to report clinical trial results on ClinicalTrials.gov: a cohort study - The Lancet